Anything Goes! Scratch built Harrison
- Thread starter mrpat
- Start date
Can you explain how you zinc plate? Great informative thread.
Zinc Plating At Home
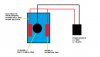
This is how I have my setup. I'm not a chemist nor do I work as a plater so I really don't know what I'm doing but I have done a lot of reading and I have proven that this does work. Play at your own risk!
The Electrolyte:
These are ratios, so I would guess that you could make this in larger batches if you wanted to:
Stir until dissolved
½ gallon distilled white vinegar
½ cup sugar
½ cup kosher salt
Power:
There are plenty of power options out there, but I personally found the phone charger option the best since I had several in an old box that aren't being used any more.
1. You can use an old cell phone charger as your power source which is what I've done. Just need to know which is positive and which is negative.
2. You can also use a 12v car battery/motorcycle battery
3. You can use a couple of 1.5v throwaway flashlight batteries – (Not sure how long they last)
You can use an older battery charger. I tried using my newer electronic protected charger which didn't work because it didn't detect a battery to charge so it shut itself off.
Other Supplies:
1. Zinc Anodes (I bought 2 chunk from Ebay for 7 bucks shipped)
2. Copper wire (thicker like 14 awg)
3. Alligator clips (Not necessary but makes it easier)
4. Wood dowel or other non conductive stick (to put across pail to hang parts from)
5. Plastic bucket for the electrolyte. I actually used a plastic apple juice bottle that I cut the top off for my tank, so it cost me nothing.
The Setup:
Put Electrolyte in the plastic bucket
Hang the Zinc Pieces over the side and ¾ submerged in the solution (I drilled a hole in the zinc and threaded a piece of the copper wire through the hole and bent a hook on the other side to hang from the outside of the bucket)
Connect the zinc pieces with a piece of the copper wire. Let zinc sit in solution for a few hours to leach some of the zinc into the electrolyte solution
Place the non conductive dowel across the bucket and hang piece to be plated from copper wire, submerging the whole part
Attach positive wire to one of the zinc pieces or copper wire connecting them and connect negative to the part to be plated being careful not to let anything from the positive touch anything from the negative.
Attach the power supply.
Bubbles should start forming and bubbling up on the part to be plated.
After about 5 minutes, disconnect the power and take part out of the bath.
Part should be a dull blueish gray color. Give the part a rinse and use a wire brush and scrub the part and it should be a bright zinc color.
**Note:
If the finished part right out of the solution looks very dark and almost sooty, this may be caused by too much current. I had this problem and corrected it by attaching a car light bulb (1157 or another similar 12v bulb) in line on the negative wire. This made the bulb glow a little bit and created just enough resistance.
Here is a pic of the rims and tires I was able to find. They need blasted and repainted, but they're a great start.

The pic below is of the sprocket and brake. Looks old and used, but it's a NOS setup. I need to do a little work to make it fit the wheels. Wheels have 5 mounting holes and the sprocket only has 4. I'll make a template and drill some new holes.


The pic below is of the sprocket and brake. Looks old and used, but it's a NOS setup. I need to do a little work to make it fit the wheels. Wheels have 5 mounting holes and the sprocket only has 4. I'll make a template and drill some new holes.

Living through the Polar vortex!! I I think im going to try that plating trick!
Last edited:
This is the one tri-star I have. Got the tire mounted (after snowblowing for an hour).
View attachment 239305





